scan from top to bottom, or left to right) look for anything that is not C-C single bond in a systematic manner (ie.Commas are used to separate the numbers for the same substituent and a dash separated the numbers from the name.Are you expected to determine the number of functional groups present in a complex compound? Or perhaps you need to identify the functional groups that are present in a given compound? I have a few tips to share: The position of attachment of each substituent is identified by the number of the carbon atom to which it is attached. Number the carbon atoms on the backbone by counting from the end nearest the substituents.If there is more than one of the same substituent, use prefixes di-, tri-, tetra-, etc to indicate the number of them. When more than one substituent is present, either on the same carbon atom or on different carbon atoms, list them alphabetically. Use the positions and names of the substituents that replace the hydrogen as prefixes.Find the longest chain of carbon atoms (the backbone) and name the atom as a derivative of the alkane with that number of carbons.Thesetwo molecules are saidto be structural isomers.īecause of multiple substitution possibilities,a naming convention has been developed to name suchderivatives.īecause of the multitude of ways organic molecules may be formed, a set of rules for naming alkane derivatives is a part of the IUPAC system for organic compounds. It has the same molecular formula as butane. For example, if a methyl group is substituted for for one the hydrogens on the center carbon of a propane molecule, the result is called methylpropane. If the substituent is an alkyl group, then the derivative will have the same empirical formula as a larger alkane, so the empirical formula for an organic compound is insufficient to identify it. Alkane derivatives can be formedby substituting an alkyl group for one of the hydrogens.Īlmost an unlimited number of derivatives can be made from the alkanes since any hydrogen can be substituted by an alkyl group, a halide, etc. Methane becomes a methyl group, ethane an ethyl group, etc.
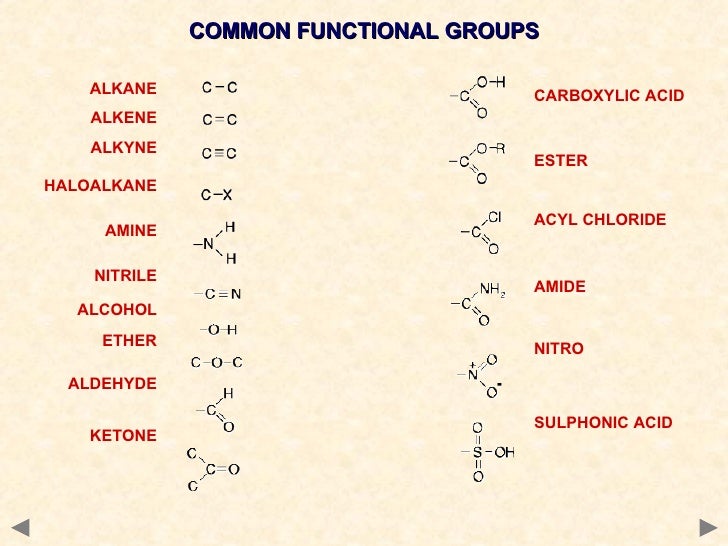
Alkyl groups are named by dropping the -ane suffix of the alkanes and adding the suffix -yl. If a hydrogen is removed from an alkane, it can be used as a substituent functional group called an alkyl group. Alkanes with higher values of n are found in diesel fuel, fuel oil, petroleum jelly, paraffin wax, motor oils, and for the highest values of n, asphalt.Īlkane derivatives are used in hundreds of products such as plastics, paints, drugs, cosmetics, detergents, insedticides, etc., so the fossil fuel resource from which we obtain the alkanes is much too valuable to burn it all as a motor fuel. Above n=17 they are solids at room temperature.
Kerosene contains alkanes from about n=10 to n=16. Gasoline is a mixture of alkanes from pentane up to about decane.

Alkanes from pentane up to around C 17H 36 are liquids. Methane, ethane, propane and butane are gases and used directly as fuels. The alkanes are highly combustible and are valuable as clean fuels, burning to form water and carbon dioxide. Alkyl groups are used as substituents, and alkane derivatives have many applications. Past this number of carbons, the -ane suffix is retained and the number prefixes penta-, hexa-, hept-, oct-, non-, dec-, etc are used. The first four alkanes are methane, ethane, propane, and butane with the Lewis symbols shown below. This gives them a general formula C nH 2n+2. They are called saturated hydrocarbons because there is a hydrogen in every possible location. Hydrocarbons which contain only single bonds are called alkanes.
